Reliable, experimentally determined partition coefficient P (logP) for most drugs are often unavailable in the literature. Many values are from in silico predictions and may not accurately reflect drug lipophilicity.

An Introduction to Mathematical Modeling in Drug Development using GastroPlus® and DILIsym®
Introduction to drug development
Introduction to PBPK modeling
Introduction to QSP/QST modeling

Halloysite nanotubes-cellulose ether based biocomposite matrix, a potential sustained release system for BCS class I drug verapamil hydrochloride: Compression characterization, in-vitro release kinetics, and in-vivo mechanistic physiologically based pharmacokinetic modeling studies
This study investigated the ability of natural nanotubular clay mineral (Halloysite) and cellulose ether based biocomposite matrix as a controlled release...
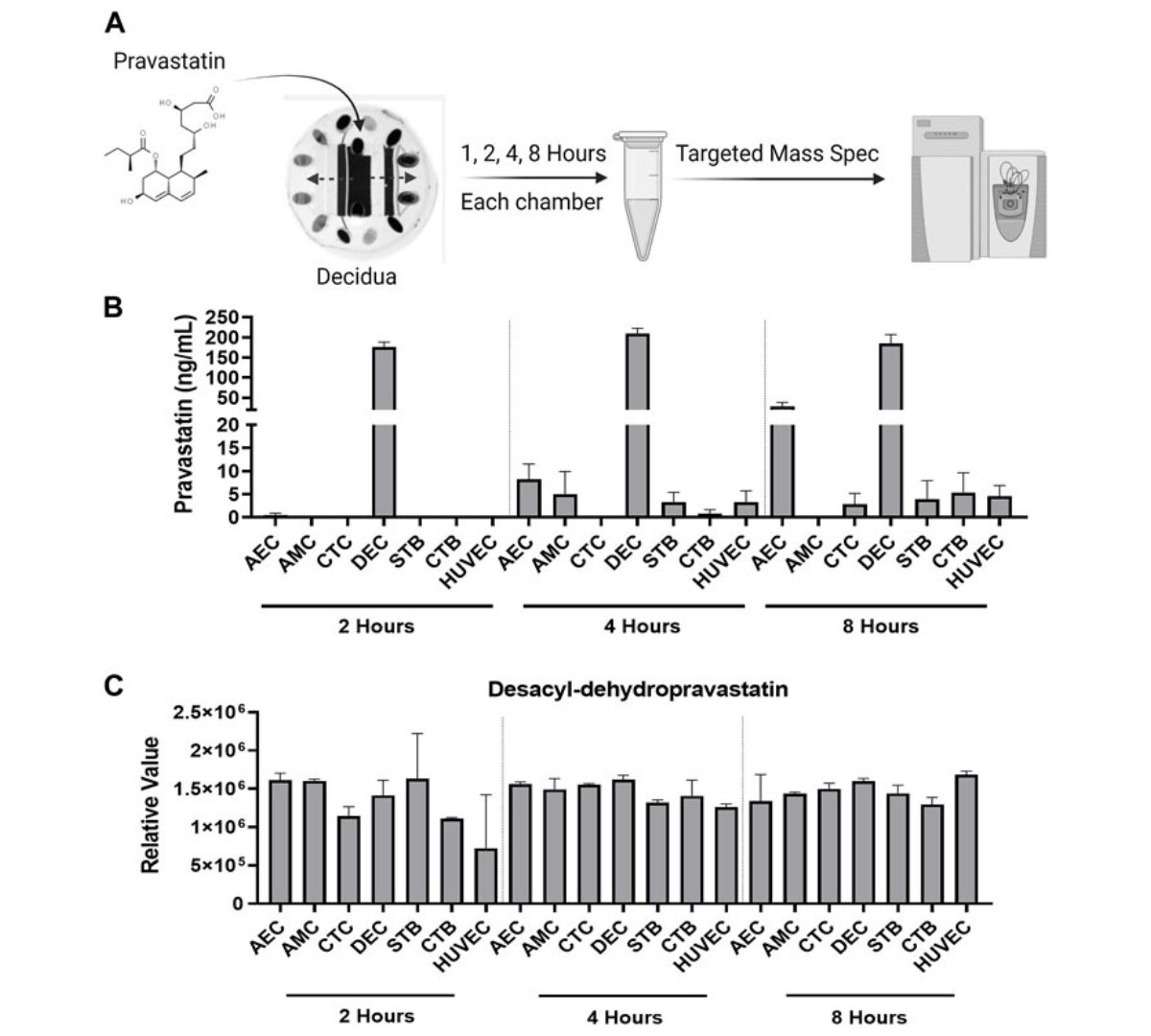
Microfluidic technology and simulation models in studying pharmacokinetics during pregnancy
Preterm birth rates and maternal and neonatal mortality remain concerning global health issues, necessitating improved strategies for testing therapeutic compounds during pregnancy.
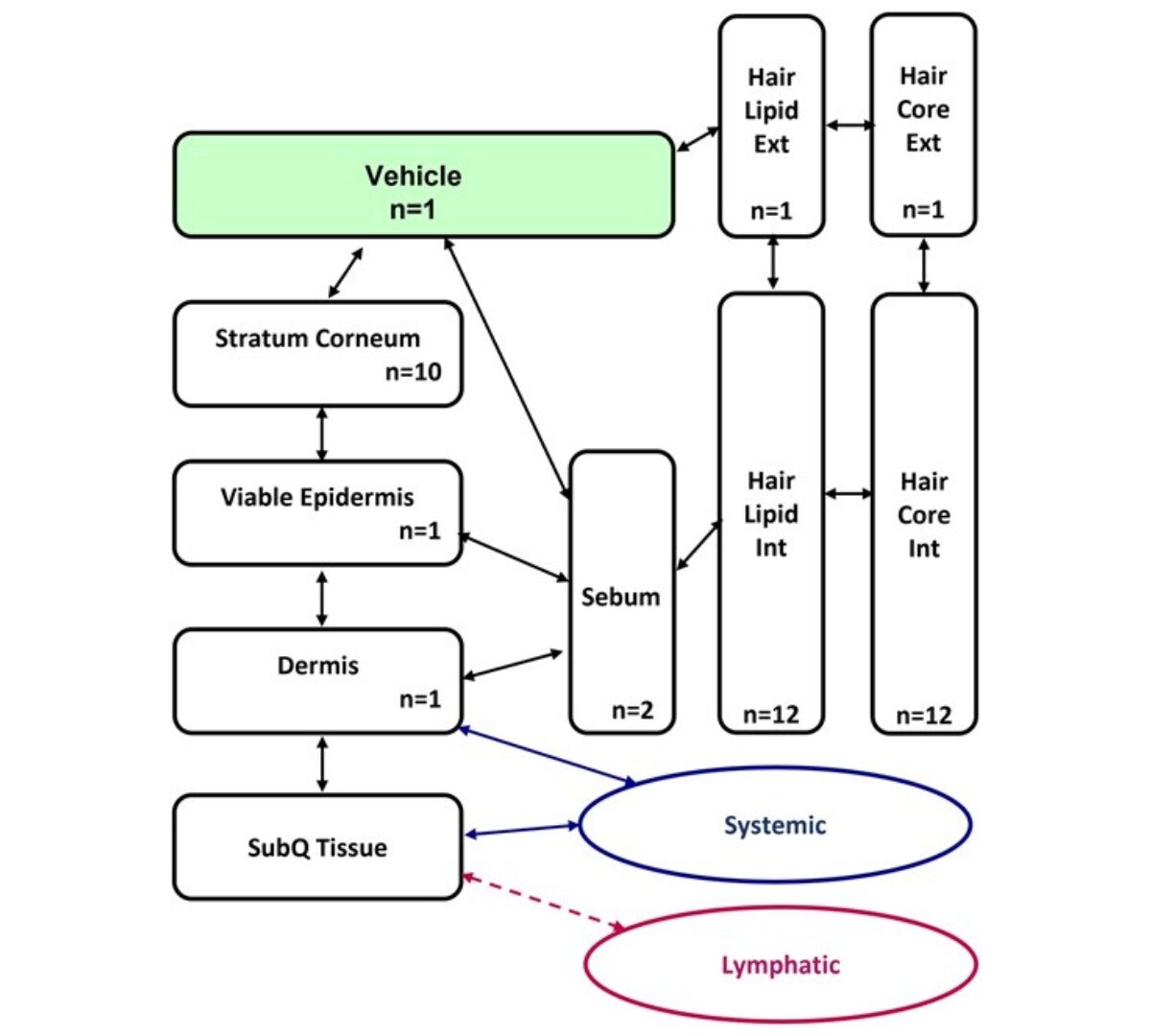
ADME characterization and PBK model development of 3 highly protein-bound UV filters through topical application
Estimating human exposure in the safety assessment of chemicals is crucial. Physiologically based kinetic (PBK) models which combine information on exposure, physiology, and chemical properties...

AIDD, an interactive AI-driven drug design system that uses molecular evolution and mechanistic pharmacokinetic simulation to optimize multiple property objectives simultaneously
Computer-aided drug design has advanced rapidly in recent years, and multiple instances of in silico designed molecules advancing to the clinic have...

Researchers Use the Simulations Plus DILIsym® Platform to Predict Safety of Combined CBD and VPA Treatment
Elevated ALT levels during clinical trials led researchers to use quantitative systems toxicology (QST) software to better understand potential underlying mechanisms
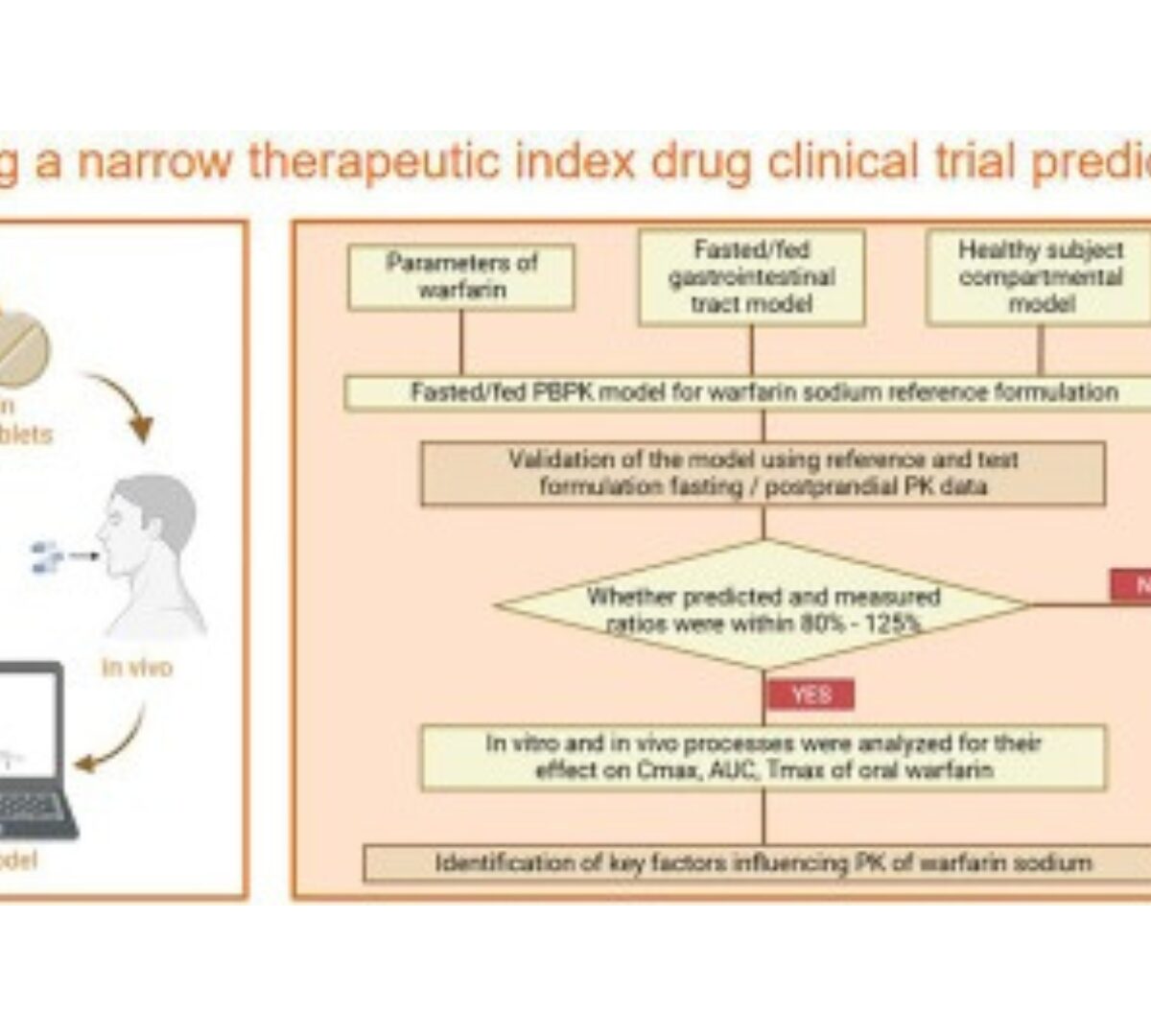
Predicting bioequivalence and developing dissolution bioequivalence safe space in vitro for warfarin using a Physiologically-Based pharmacokinetic absorption model
Bioequivalence (BE) studies support the approval and clinical use of both new and generic drug products.

Training the next generation of pharmacometric modelers: a multisector perspective
The current demand for pharmacometricians outmatches the supply provided by academic institutions and considerable investments are made to develop the competencies of these scientists on-the-job.

Physiologically based pharmacokinetic model combined with reverse dose method to study the nephrotoxic tolerance dose of tacrolimus
Nephrotoxicity is the most common side effect that severely limits the clinical application of tacrolimus (TAC), an immunosuppressive agent used in kidney transplant patients.

The In Vitro, In Vivo, and PBPK Evaluation of a Novel Lung-Targeted Cardiac-Safe Hydroxychloroquine Inhalation Aerogel
Hydroxychloroquine (HCQ) was repurposed for COVID-19 treatment. Subtherapeutic HCQ lung levels and cardiac toxicity of oral HCQ were overcome...

August 2023 GastroPlus Newsletter
GastroPlus® Newsletter August 2023

PB2205: A Mechanistic Absorption and Pharmacokinetic Model of Covalent BTK Inhibitor TL-895: Influence of Food and Acid Reducing Agents
L-895 is a highly potent, orally available, selective, covalent inhibitor of Bruton tyrosine kinase (BTK) and bone marrow tyrosine kinase X-linked (BMX)...
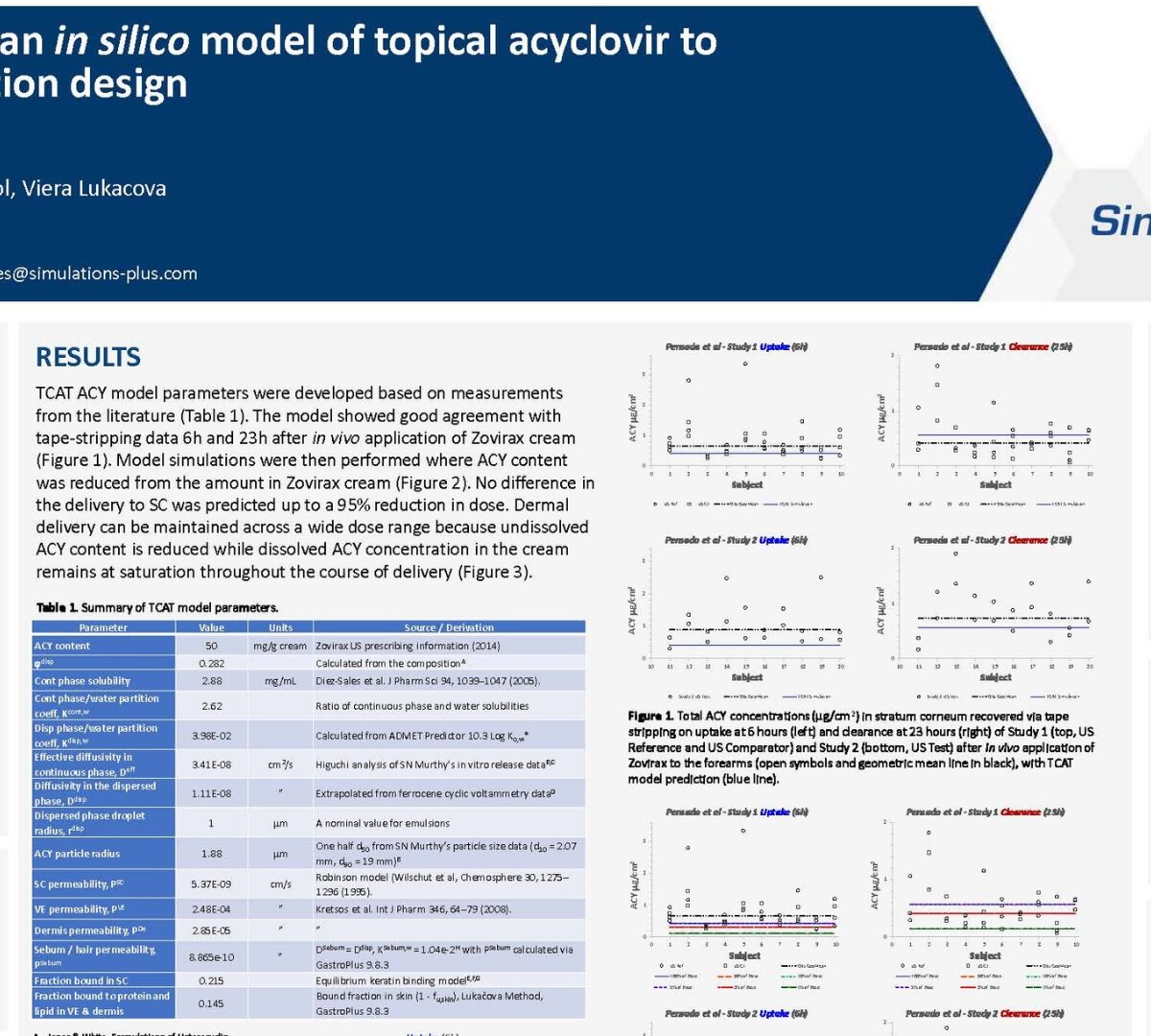
Development of an in silico model of topical acyclovir to explore formulation design
Acyclovir (ACY) creams are used for local treatment of HSV-1 infections in the basal epidermis.

Quantitative Systems Toxicology identifies independent mechanisms for hepatotoxicity and bilirubin elevations due to AKR1C3 Inhibitor BAY1128688
BAY1128688 is a selective inhibitor of AKR1C3, investigated recently in a trial that was prematurely terminated due to drug-induced liver injury. These...

Assessing Liver Effects of Cannabidiol and Valproate Alone and in Combination Using Quantitative Systems Toxicology
In clinical trials of cannabidiol (CBD) for the treatment of seizures in patients with Dravet syndrome, Lennox-Gastaut syndrome, and tuberous sclerosis complex, elevations in serum alanine...

Discovering Negative Allosteric Modulators of Frizzled 4 Receptor Using the NPASS Database
The class F G protein-coupled receptors (GPCRs) consists of the Smoothened receptor (SMO) and 10 Frizzled receptors (FZDs).

August 2023 News/Events
Quickening the pace of drug discovery with AI

The Organophosphate Esters Used as Flame Retardants and Plasticizers Affect H295R Adrenal Cell Phenotypes and Functions
Adverse effects associated with exposure to brominated flame retardants have led to regulations for their use and their replacement with organophosphate esters (OPEs).

In Silico Modeling Approaches Coupled with In Vitro Characterization in Predicting In Vivo Performance of Drug Delivery System Formulations
Optimization of the in vivo performance of dosage forms in humans is essential in developing not only conventional formulations but also drug delivery system (DDS) formulations.
