
A combination of pharmacophore modeling, molecular docking, and virtual screening for P2Y12 receptor antagonists from Chinese herbs
P2Y12, a member of the G-protein-coupled receptors, is associated with abnormal platelet aggregation, a condition that contributes to thrombus formation.

Simulations Plus Sets Date for 4th Quarter and Fiscal Year 2014 Earnings Release and Conference Call
Conference Call to be on Tuesday, November 25, 2014, at 4:15 PM EST
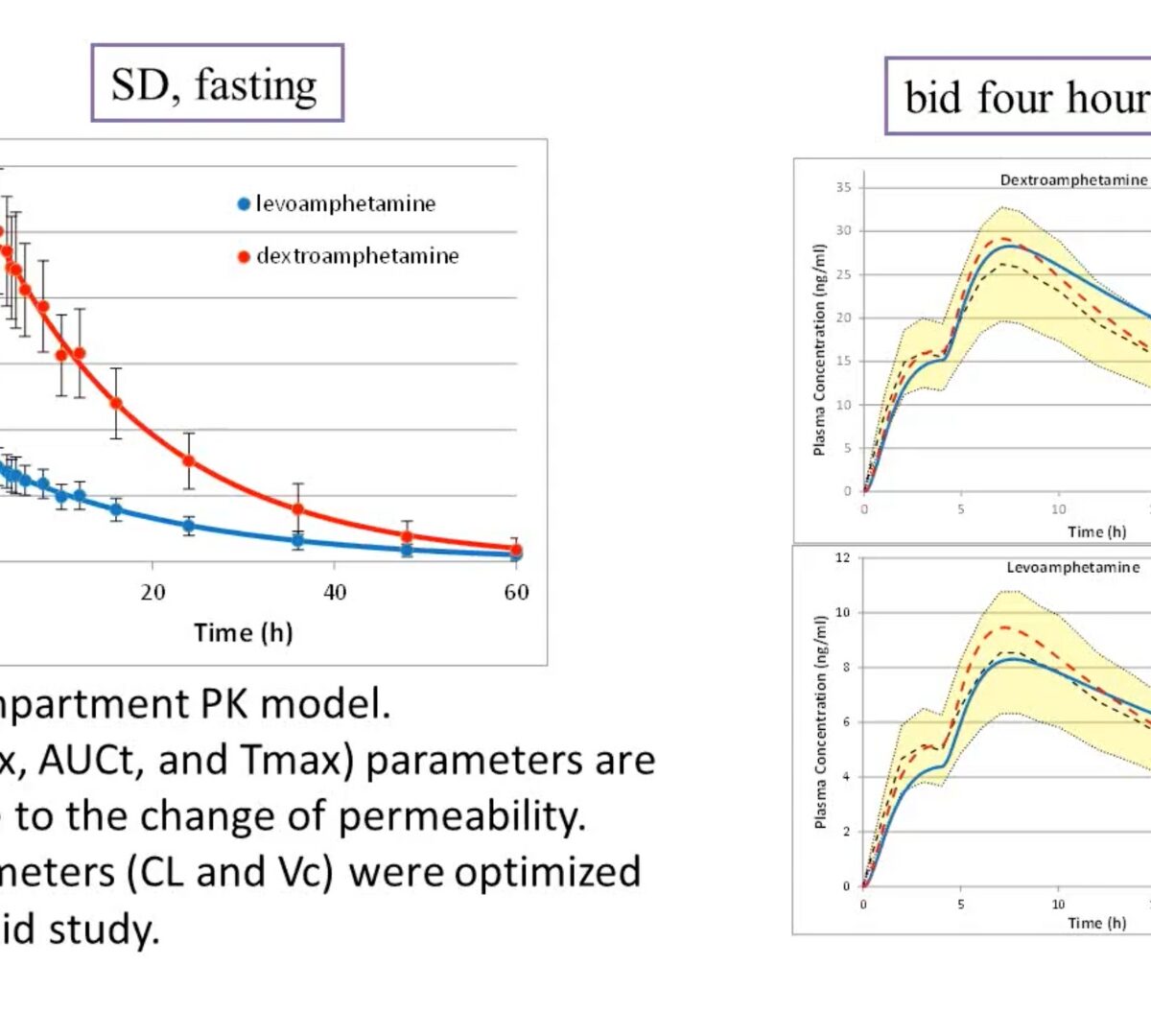
Application of PBPK Modeling in Generic Drug Evaluation
This GastroPlus™ User Group/FDA webinar provides a few examples of modeling and simulation in the OGD for the purpose of addressing biopharmaceutical performance questions for orally administered generic drug products. View slides from this webinar.

Prediction and Comparison of Drug likeliness properties of Primaquine and its structural analogues using In-Silico ADME and Toxicity Prediction Tools
Five primaquine (PQ) analogues have been isolated by peroxydisulfate oxidation and were tested for antimalarial activity against Plasmodium yoelli infected mice.

Three-Dimensional Quantitative Structure-Activity Relationship Analysis for Human Pregnane X Receptor for the Prediction of CYP3A4 Induction in Human Hepatocytes: Structure-Based Comparative Molecular Field Analysis
The pregnane X receptor [PXR (NR1I2)] induces the expression of xenobiotic metabolic genes and transporter genes. In this study, we aimed to establish a computational method for...

Exploring BSEP inhibition-mediated toxicity with a mechanistic model of drug-induced liver injury
Inhibition of the bile salt export pump (BSEP) has been linked to incidence of drug-induced liver injury (DILI), presumably by the accumulation of toxic bile acids in the liver.

BU08073 a Buprenorphine Analog with Partial Agonist Activity at mu Receptors in vitro but Long-Lasting Opioid Antagonist Activity in vivo in Mice
Buprenorphine is a potent analgesic with high affinity at μ, δ and κ and moderate affinity at nociceptin opioid (NOP) receptors.

Novel Physiologically-Based Oral Cavity Model and Its Application for Projection of Clinical Pharmacokinetics of Intermezzo® Sublingual Tablets
Intraoral (IO) delivery refers to an alternative administration route that intends to deliver the drug substance through oral mucosa. The intraoral route provides several advantages over conventional...

Simulations Plus Announces Changes in Board of Directors
Dr. John Paglia, Ph.D., CFA, CPA to Replace Mr. Harold Wayne Rosenberger Retiring after 7 Years on Board

Prediction of Acyclovir Pharmacokinetics in Pediatric Populations using a Physiologically Based Pharmacokinetic (PBPK) Model
To develop a PBPK model for acyclovir incorporating processes affecting the drug's absorption and distribution after i.v. administration of acyclovir (ACV) as well as its in vivo formation after p.o. admin…

Mechanistic Nucleation and Growth Can Predict Nonlinear Dose-Dependent Precipitation of Enabled Fomulations of Nimodipine in GastroPlus™
Simulation of precipitation can be important for accurate prediction of the absorption of many oral formulations. Amorphous solid dispersions, salt forms, cocrystals, solutions, and nanoparticles all can…

Physiologically Based Pharmacokinetic Modeling of Buspirone and the Effect of Liver Cirrhosis on Its Deposition
The aim of this work was to develop a physiologically based pharmacokinetic (PBPK) model of buspirone following oral administrations in healthy volunteers, and to extend this model to predict the…

Novel Skin PBPK model in Action: Clindamycin and Tazarotene Modeling
Skin is the major organ in the human body with a complex barrier that serves as protection from the external environment. Predicting dermal and systemic exposures of drug following topical application...

Population Modeling Is a Critical Element of Bridging Pharmacokinetic Data From Dried Blood Spot and Plasma Across Clinical Programs
To demonstrate through two case studies how population pharmacokinetic (PK) modeling should be leveraged for bridging plasma and dried blood spot (DBS) PK data across clinical development programs.

Evaluation of a Three Compartment In Vitro Gastrointestinal Simulator Dissolution Apparatus to Predict In Vivo Dissolution
In vitro dissolution tests are performed for new formulations to evaluate in vivo performance, which is affected by the change of gastrointestinal (GI) physiology, in the GI tract.

Elucidation of Arctigenin Pharmacokinetics After Intravenous and Oral Administrations in Rats: Integration of In Vitro and In Vivo Findings via Semi-mechanistic Pharmacokinetic Modeling
Although arctigenin (AR) has attracted substantial research interests due to its promising and diverse therapeutic effects, studies regarding its biotransformation were limited.

Preparation and Evaluation of High Dispersion Stable Nanocrystal Formulation of Poorly Water-Soluble Compounds by Using Povacoat
In this study, we reported the application of Povacoat®, a hydrophilic polyvinylalcohol copolymer, as a dispersion stabilizer of nanoparticles of poorly water-soluble compounds.

A new in vitro system for evaluation of passive intestinal drug absorption: Establishment of a double artificial membrane permeation assay
The aim of this present study was to establish a new in vitro assay, double artificial membrane permeation assay (DAMPA), to evaluate the human intestinal permeability of drugs.

The Poorly Membrane Permeable Antipsychotic Drugs Amisulpride and Sulpiride Are Substrates of the Organic Cation Transporters from the SLC22 Family
Variations in influx transport at the blood-brain barrier might affect the concentration of psychotropic drugs at their site of action and as a consequence might alter therapy response.
