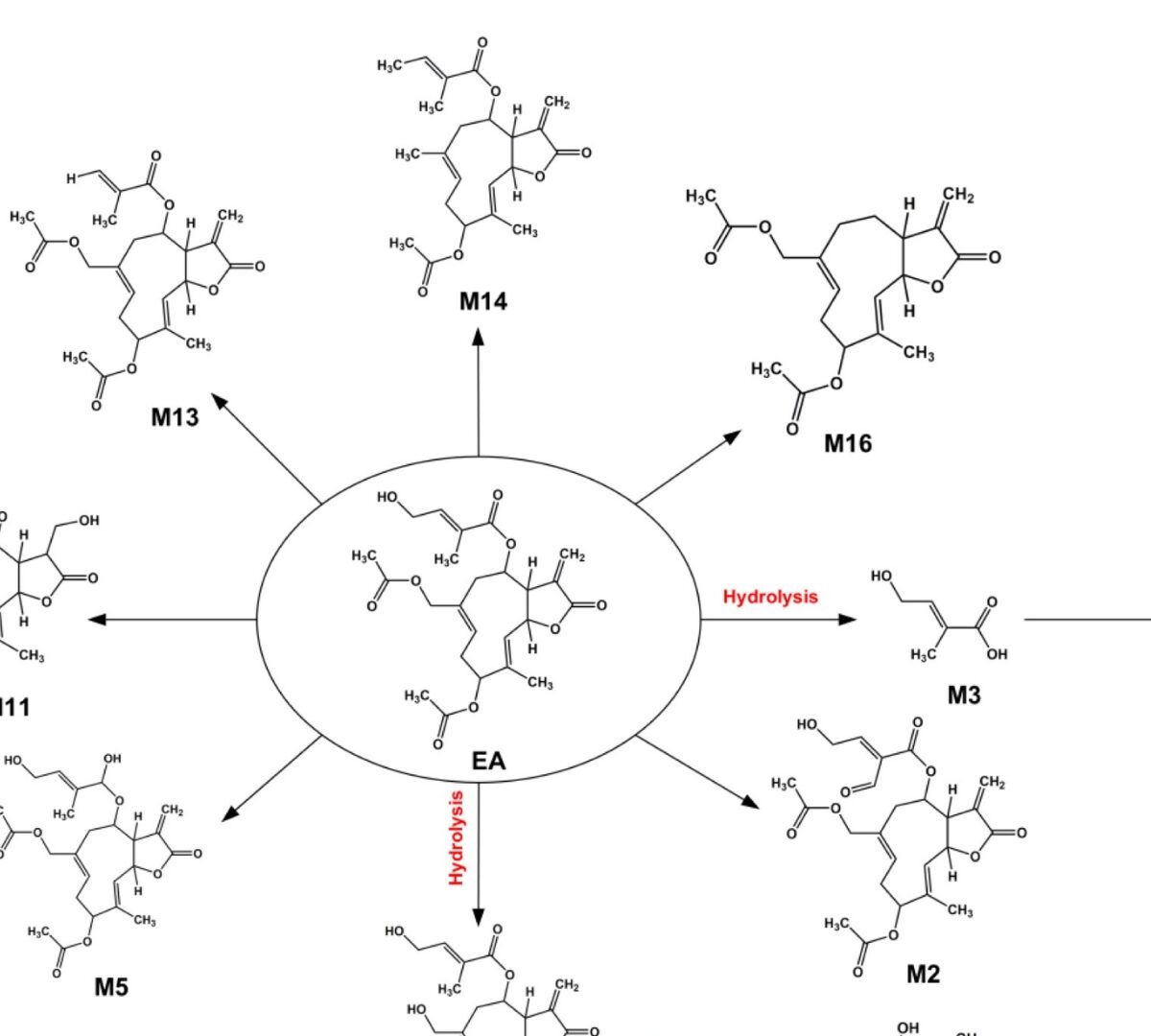
Eupalinolide A (EA, Z-configuration) and Eupalinolide B (EB, E-configuration) are cis-trans isomeric sesquiterpenoid monomers isolated from Eupatorium lindleyanum DC. (Asteraceae).

Automated Concentration-QT data preparation, model selection and reporting in R
Since the publication of the ICH E14 guidance in 2015, QT interval prolongation as-sessment can be carried out with a concentration-QTc modeling approach as part of single- or mul-tiple- dose escalation studies, instead of conducting a thorough QT/QTc study.
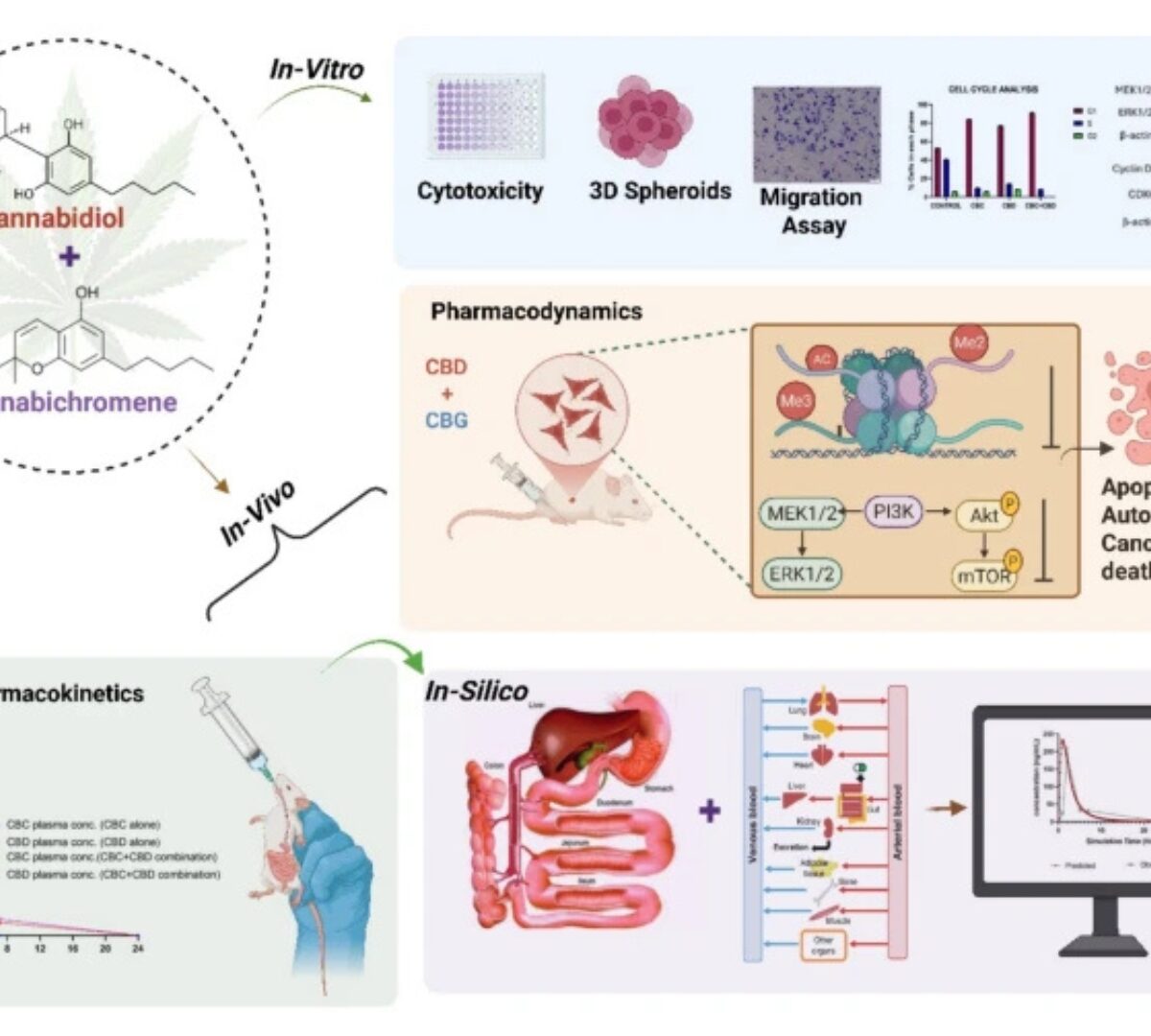
Pharmacokinetic Studies and Synergistic Antitumor Effects of Cannabichromene and Cannabidiol in Drug-Resistant Breast Cancers
Triple-negative breast cancer (TNBC) is highly aggressive with limited treatment options, and resistance to doxorubicin (DOX) further compromises outcomes.

Building Confidence in Physiologically BasedPharmacokinetic Modeling of CYP3A InductionMediated by Rifampin: An Industry Perspective
Physiologically- based pharmacokinetic (PBPK) modeling offers a viable approach to predict induction drug–druginteractions (DDIs) with the potential to streamline or reduce clinical trial burden if predictions can be made withsufficient confidence.

High-Throughput Physiologically Based Pharmacokinetic Model for Rodent Pharmacokinetics Prediction Using Machine Learning-Predicted Inputs and a Large In Vivo Pharmacokinetics Data Set
Accurate prediction of the pharmacokinetic (PK) properties of small-molecule drug candidates is a critical aspect of pharmaceutical research.

Design, Synthesis, and Pharmacological Evaluation of Metabolically Stable Apelin Receptor Antagonists with Improved In Vivo Exposure and Efficacy in Ovarian Cancer
The apelin receptor (APJ) has emerged as a potential novel therapeutic target in cancer due to its role in regulating cell proliferation, angiogenesis, and metastasis.

Investigating the SNAC-To-Drug Ratio for the Oral Absorption of Cyanocobalamin in Rats
The oral route of administration continues to be the preferred and most convenient method for drug delivery among both patients and healthcare professionals.

Novel Descriptors and Models for More Accurate ADME Predictions of Beyond Rule of Five Molecules
This study addresses the critical need for improved physicochemical descriptors and predictive models tailored to beyond Rule of Five (bRo5) compounds, including PROTACs and cyclic peptides. B

Development of a Pregnancy-Specific Physiologically Based Pharmacokinetics (PBPK) Model for Aspirin
Aspirin is one of the most commonly used medications in pregnancy, particularly for the prevention of hypertensive disorders.

Quantitative Systems Toxicology Modeling with DILIsym to Support Phase 3 Dose Selection for Fezolinetant
Fezolinetant is a first-in-class, selective, non-hormonal, neurokinin 3 receptor antagonist that is approved for the treatment of moderate to severe vasomotor symptoms due to menopause.

Fabrication of Second-Generation Acalabrutinib Nanocrystals by Employing the Nano-Edge Method for Improving the Physico-Chemical Properties and Forecasting their In-Silico Pharmacokinetic Behaviour
Acalabrutinib (ACL) is approved by the USFDA and classified as a BCS class II drug, primarily used for treating chronic lymphocytic leukaemia.

Unveiling Anticancer Potential of Imidazo-Pyridine/Pyrazine-Fused bicyclic Heterocycles: Insights From in silico and in vitro Studies
Cancer is a significant global health challenge and the second leading cause of death worldwide, responsible for millions of fatalities annually.

Physiologically Based Pharmacokinetic Model for Clinical Translation and Prediction of Drug Interaction of the Major Kratom Alkaloid, Mitragynine
The opioid crisis presents a significant public health issue and underscores the urgency of developing effective treatments for opioid use disorder (OUD).

Supramolecular Organogels Based on Cinnarizine as a Potential Gastroretentive System: In Vitro and In Silico Simulations
Gastroretentive systems are an interesting option for enhancing the bioavailability of weak bases and poorly soluble drugs.

Exploration of IVIVC Deconvolution Methods in a PBPK Platform: Case Example with Tofacitinib
Modified-release (MR) drug products are designed to provide controlled drug delivery over time, offering therapeutic and compliance advantages.

Oral Pro‑Transferosome Tablets of Carica papaya Leaf Extract: Box–Behnken Optimization and In Silico Mechanistic Insights for Dengue‑Associated Thrombocytopenia
Carica papaya L. leaf extract has long been recognized for its therapeutic potential in dengue-associated thrombocytopenia through enhancement of platelet production and inhibition of dengue viral proteases.

Comprehensive PBPK Evaluation of Phenytoin and Indomethacin: Dose, Age, Pregnancy and Drug–Drug Interaction Insights
Understanding the pharmacokinetics (PK) of antiepileptic and anti-inflammatory drugs under different physiological conditions is essential for optimizing therapy.

Descriptor-First Approach for ADMET Prediction in the PolarisHub Antiviral Challenge
The prediction of absorption, distribution, metabolism, excretion, and toxicity (ADMET) properties remains a central bottleneck in small-molecule discovery.

Mechanistic Modeling of Intramuscular Administration of a Long-acting Injectable Accounting for Tissue Response At the Depot Site
The tissue response to long-acting injectables (LAIs) suspension injection may impact the product in vivo performance.

Model-Based Meta-Analysis With MonolixSuite: A Tutorial for Longitudinal Categorical and Continuous Data
Model-based meta-analysis (MBMA) informs key drug development decisions by integrating data, published or unpublished, from multiple studies.